Abstract
Ryanodine receptors (RyRs), intracellular calcium release channels required for cardiac and skeletal muscle contraction, are macromolecular complexes that include kinases and phosphatases. Phosphorylation/dephosphorylation plays a key role in regulating the function of many ion channels, including RyRs. However, the mechanism by which kinases and phosphatases are targeted to ion channels is not well understood. We have identified a novel mechanism involved in the formation of ion channel macromolecular complexes: kinase and phosphatase targeting proteins binding to ion channels via leucine/isoleucine zipper (LZ) motifs. Activation of kinases and phosphatases bound to RyR2 via LZs regulates phosphorylation of the channel, and disruption of kinase binding via LZ motifs prevents phosphorylation of RyR2. Elucidation of this new role for LZs in ion channel macromolecular complexes now permits: (a) rapid mapping of kinase and phosphatase targeting protein binding sites on ion channels; (b) predicting which kinases and phosphatases are likely to regulate a given ion channel; (c) rapid identification of novel kinase and phosphatase targeting proteins; and (d) tools for dissecting the role of kinases and phosphatases as modulators of ion channel function.
Keywords: leucine zipper, ryanodine receptor, calcium channel, phosphorylation, phosphatase
CoPilot on leucine/isoleucine zippers
The leucine/isoleucine zipper (LZ) is a type of protein structural motif that facilitates protein-protein interactions. It is characterized by the presence of leucine or isoleucine residues at every seventh position in an alpha-helix, which allows the helices to dimerize and form a stable coiled-coil structure12.
This motif is commonly found in transcription factors, where it plays a crucial role in DNA binding and gene regulation3. The leucine zipper forms a dimeric parallel coiled-coil, but can also oligomerize to form trimers, tetramers, or pentamers depending on the specific amino acids involved2.
Learn more
- 1nature.com
- 2 acsu.buffalo.edu
- 3 en.wikipedia.org
- 4 link.springer.com
- 5 academic.oup.com
- 6 microbialcellfactories.biomedcentral.com
The leucine/isoleucine zipper (LZ) motif interacts with DNA primarily through its role in transcription factors. Here’s how it works:
- Dimerization: The LZ motif allows two protein molecules to dimerize, forming a stable coiled-coil structure. This dimerization is crucial because it positions the DNA-binding domains of the transcription factors in the correct orientation to interact with DNA.
- DNA Binding: Once dimerized, the transcription factors can bind to specific DNA sequences. The DNA-binding domains, often rich in basic amino acids, interact with the major groove of the DNA helix. This interaction is highly specific, allowing the transcription factors to recognize and bind to particular DNA sequences.
- Gene Regulation: By binding to DNA, the LZ-containing transcription factors can regulate the expression of target genes. They can either activate or repress transcription depending on the context and the specific transcription factor involved.
The leucine zipper itself does not directly interact with DNA; instead, it facilitates the dimerization necessary for the DNA-binding domains to function effectively.
There are several well-known transcription factors that contain the leucine/isoleucine zipper (LZ) motif. Here are a few examples:
- c-Fos and c-Jun: These proteins form the AP-1 transcription factor complex, which is involved in regulating gene expression in response to a variety of stimuli, including cytokines, growth factors, stress, and bacterial and viral infections1.
- Myc family: This includes c-Myc, N-Myc, and L-Myc, which are crucial for cell cycle progression, apoptosis, and cellular transformation. Overexpression or mutation of these proteins is often associated with cancer1.
- Sterol Regulatory Element-Binding Proteins (SREBPs): These transcription factors regulate lipid homeostasis by controlling the expression of genes required for the synthesis and uptake of cholesterol, fatty acids, and triglycerides2.
- Microphthalmia-associated Transcription Factor (MITF): This factor is important for the development and function of melanocytes, and mutations in MITF can lead to various pigmentation disorders2.
- Nuclear Factor Interleukin 3 Regulated (NFIL3): This transcription repressor plays multiple roles in regulating various biological processes, including immune responses1.
These transcription factors utilize the LZ motif to dimerize and bind to specific DNA sequences, thereby regulating the expression of target genes.
Learn more
Wikipedia on ryanodine receptors
The ryanodine receptors are named after the plant alkaloid ryanodine which shows a high affinity to them.
Ryania is a genus of flowering plants in the family Salicaceae; it was previously listed in the now defunct family Flacourtiaceae. The Salicaceae are the willow family of flowering plants. The traditional family (Salicaceae sensu stricto) included the willows, poplars, aspens, and cottonwoods. Genetic studies summarized by the Angiosperm Phylogeny Group (APG) have greatly expanded the circumscription of the family to contain 56 genera and about 1220 species, including the tropical Scyphostegiaceae and many of the former Flacourtiaceae. The Flacourtiaceae is a defunct family of flowering plants whose former members have been scattered to various families, mostly to the Achariaceae and Salicaceae. It was so vaguely defined that hardly anything seemed out of place there and it became a dumping ground for odd and anomalous genera, gradually making the family even more heterogeneous. In 1975, Hermann Sleumer noted that “Flacourtiaceae as a family is a fiction; only the tribes are homogeneous.” In Cronquist’s classification, the Flacourtiaceae included 79–89 genera and 800–1000 species. Of these, many, including the type genus Flacourtia, have now been transferred to the Salicaceae in the molecular phylogeny-based classification, known as the APG IV system, established by the Angiosperm Phylogeny Group. In the list below, the Salicaceae are circumscribed broadly. Some taxonomists further divide the Salicaceae sensu lato into three families: Salicaceae sensu stricto, Scyphostegiaceae, and Samydaceae, or into three subfamilies. Ryania is currently listed Samydaceae.
- Roskov Y.; Kunze T.; Orrell T.; Abucay L.; Paglinawan L.; Culham A.; Bailly N.; Kirk P.; Bourgoin T.; Baillargeon G.; Decock W.; De Wever A. (2014). Didžiulis V. (ed.). “Species 2000 & ITIS Catalogue of Life: 2014 Annual Checklist”. Species 2000: Reading, UK. Retrieved 26 May 2014.
- Chase, Mark W.; Sue Zmarzty; M. Dolores Lledó; Kenneth J. Wurdack; Susan M. Swensen; Michael F. Fay (2002). “When in doubt, put it in Flacourtiaceae: a molecular phylogenetic analysis based on plastid rbcL DNA sequences”. Kew Bulletin. 57 (1): 141–181. Bibcode:2002KewBu..57..141C. doi:10.2307/4110825. JSTOR 4110825.
- Christenhusz, M. J. M. & Byng, J. W. (2016). “The number of known plants species in the world and its annual increase”. Phytotaxa. 261 (3). Magnolia Press: 201–217. doi:10.11646/phytotaxa.261.3.1.
- Stevens, P. F. (2001 onwards). Angiosperm Phylogeny Website. Version 9, June 2008 (and more or less continuously updated since).
- Miller, Regis B. (1975). “Systematic anatomy of the xylem and comments on the relationships of Flacourtiaceae”. Journal of the Arnold Arboretum. 56 (1): 79. doi:10.5962/p.185846. S2CID 108459529.
- Lemke, David (1988). “A synopsis of Flacourtiaceae”. Aliso. 12 (1): 29–43. doi:10.5642/aliso.19881201.05. S2CID 87472599. Retrieved 11 July 2018.
- Wurdack, Kenneth J.; Charles C. Davis (2009). “Malpighiales phylogenetics: Gaining ground on one of the most recalcitrant clades in the angiosperm tree of life”. American Journal of Botany. 96 (8): 1551–1570. doi:10.3732/ajb.0800207. PMID 21628300. S2CID 23284896.
Isoforms
There are multiple isoforms of ryanodine receptors:
- RyR1 is primarily expressed in skeletal muscle
- RyR2 is primarily expressed in myocardium (heart muscle)
- RyR3 is expressed more widely, but especially in the brain.[2]
- Non-mammalian vertebrates typically express two RyR isoforms, referred to as RyR-alpha and RyR-beta.
- Many invertebrates, including the model organisms Drosophila melanogaster (fruitfly) and Caenorhabditis elegans, only have a single isoform. In non-metazoan species, calcium-release channels with sequence homology to RyRs can be found, but they are shorter than the mammalian ones and may be closer to IP3 Receptors.
Pharmacology
- Antagonists:[11]
- Ryanodine locks the RyRs at half-open state at nanomolar concentrations, yet fully closes them at micromolar concentration.
- Dantrolene the clinically used antagonist
- Ruthenium red
- The inorganic dye ammoniated ruthenium oxychloride, also known as ruthenium red, is used in histology to stain aldehyde fixed mucopolysaccharides.
- Ruthenium red (RR) has also been used as a pharmacological tool to study specific cellular mechanisms. Selectivity is a significant issue in such studies as RR is known to interact with many proteins.[ Vincent, F; Duncton, MA (2011). “TRPV4 agonists and antagonists”. Curr Top Med Chem. 11 (17): 2216–26. doi:10.2174/156802611796904861. PMID 21671873.] These include mammalian ion channels (CatSper1, TASK, RyR1, RyR2, RyR3, TRPM6, TRPM8, TRPV1, TRPV2, TRPV3, TRPV4, TRPV5, TRPV6, TRPA1, mCa1, mCa2, CALHM1[Ma, Z; Siebert, AP; Cheung, KH; Lee, RJ; Johnson, B; Cohen, AS; Vingtdeux, V; Marambaud, P; Foskett, JK (2012). “Calcium homeostasis modulator 1 (CALHM1) is the pore-forming subunit of an ion channel that mediates extracellular Ca2+ regulation of neuronal excitability”. Proc Natl Acad Sci USA. 109 (28): E1963–71. Bibcode:2012PNAS..109E1963M. doi:10.1073/pnas.1204023109. PMC 3396471. PMID 22711817.][Dreses-Werringloer, U; Vingtdeux, V; Zhao, H; Chandakkar, P; Davies, P; Marambaud, P (2013). “CALHM1 controls Ca2+-dependent MEK/ERK/RSK/MSK signaling in neurons”. J Cell Sci. 126 (Pt 5): 1199–206. doi:10.1242/jcs.117135. PMC 4481642. PMID 23345406.]) TRPP3,[Decaen, P. G.; Delling, M.; Vien, T. N.; Clapham, D. E. (2013). “Direct recording and molecular identification of the calcium channel of primary cilia”. Nature. 504 (7479): 315–318. Bibcode:2013Natur.504..315D. doi:10.1038/nature12832. PMC 4073646. PMID 24336289.] a plant ion channel, Ca2+-ATPase, mitochondrial Ca2+ uniporter,[Hajnóczky, G; Csordás, G; Das, S; Garcia-Perez, C; Saotome, M; Sinha Roy, S; Yi, M (2006). “Mitochondrial calcium signalling and cell death: approaches for assessing the role of mitochondrial Ca2+ uptake in apoptosis”. Cell Calcium. 40 (5–6): 553–60. doi:10.1016/j.ceca.2006.08.016. PMC 2692319. PMID 17074387.] tubulin, myosin light-chain phosphatase, and Ca2+ binding proteins such as calmodulin. Ruthenium red displays nanomolar potency against several of its binding partners (e.g. TRPV4, ryanodine receptors,…). For example, it is a potent inhibitor of intracellular calcium release by ryanodine receptors (Kd ~20 nM).[Tripathy, Le Xu Ashutosh; Pasek, Daniel A.; Meissner, Gerhard (1998). “Potential for Pharmacology of Ryanodine Receptor/Calcium Release Channels”. Ann NY Acad Sci. 853 (1): 130–148. Bibcode:1998NYASA.853..130T. doi:10.1111/j.1749-6632.1998.tb08262.x. PMID 10603942. S2CID 86436194. Archived from the original on 2008-04-23. Retrieved 2006-10-22.] As a TRPA1 blocker, it assists in reducing the airway inflammation caused by pepper spray.
- RR has been used on plant material since 1890 for staining pectins, mucilages, and gums. RR is a stereoselective stain for pectic acid, insofar as the staining site occurs between each monomer unit and the next adjacent neighbor.[Mariani Colombo P, Rascio N. “Ruthenium red staining for electron microscopy of plant material”. Journal of Ultrastructure Research Volume 60, Issue 2, August 1977, Pages 135–139]
- all these notes are from Ruthenium red – Wikipedia
- procaine, tetracaine, etc. (local anesthetics)
- Activators:[12]
- Agonist: 4-chloro-m-cresol and suramin are direct agonists, i.e., direct activators.
- Suramin is a medication used to treat African sleeping sickness and river blindness.[“Suramin Injection Advanced Patient Information”. Drugs.com. 3 January 2020. Retrieved 11 January 2020.][“Micromedex Detailed Drug Information for the Consumer: Suramin (Injection route)”. PubMed Health. 1 November 2016. Archived from the original on 8 September 2017.] It is the treatment of choice for sleeping sickness without central nervous system involvement.[“Our Formulary Infectious Diseases Laboratories CDC”. www.cdc.gov. 22 September 2016. Archived from the original on 16 December 2016. Retrieved 30 November 2016.] It is given by injection into a vein.
- Suramin causes a fair number of side effects.[Zuckerman JN (2002). Principles and Practice of Travel Medicine. John Wiley & Sons. p. 113. ISBN 9780471490791. Archived from the original on 30 November 2016.] Common side effects include nausea, vomiting, diarrhea, headache, skin tingling, and weakness. Sore palms of the hands and soles of the feet, trouble seeing, fever, and abdominal pain may also occur. Severe side effects may include low blood pressure, decreased level of consciousness, kidney problems, and low blood cell levels. It is unclear if it is safe when breastfeeding.[“Micromedex Detailed Drug Information for the Consumer: Suramin (Injection route)”. PubMed Health. 1 November 2016. Archived from the original on 8 September 2017.]
- Suramin was made at least as early as 1916.[Mehlhorn H (2008). Encyclopedia of Parasitology: A-M. Springer Science & Business Media. p. 475. ISBN 9783540489948. Archived from the original on 30 November 2016.] It is on the World Health Organization’s List of Essential Medicines.[World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.] In the United States it can be acquired from the Centers for Disease Control (CDC).[“Our Formulary Infectious Diseases Laboratories CDC”. www.cdc.gov. 22 September 2016. Archived from the original on 16 December 2016. Retrieved 30 November 2016.] In regions of the world where the disease is common suramin is provided for free by the World Health Organization (WHO).[“Trypanosomiasis, human African (sleeping sickness)”. World Health Organization. February 2016. Archived from the original on 4 December 2016. Retrieved 7 December 2016.]
- Suramin has been applied clinically to HIV/AIDS patients resulting in a significant number of fatal occurrences and as a result the application of this molecule was abandoned for this condition.[Kaplan LD, Wolfe PR, Volberding PA, Feorino P, Levy JA, Abrams DI, et al. (March 1987). “Lack of response to suramin in patients with AIDS and AIDS-related complex”. The American Journal of Medicine. 82 (3 Spec No): 615–620. doi:10.1016/0002-9343(87)90108-2. PMID 3548350.]
- The mechanism of action for suramin is unclear, but it is thought that parasites are able to selectively uptake suramin via receptor-mediated endocytosis of drug that is bound to low-density lipoproteins and, to a lesser extent, other serum proteins.[Phillips MA, Stanley Jr SL (2011). “Chapter 50: Chemotherapy of Protozoal Infections: Amebiasis, Giardiasis, Trichomoniasis, Trypanosomiasis, Leishmaniasis, and Other Protozoal Infections”. In Brunton LL, Chabner BA, Knollmann BC (eds.). Goodman and Gilman’s The Pharmacological Basis of Therapeutics (12th ed.). McGraw Hill. pp. 1437–1438. ISBN 9780071769396.] Once inside parasites, suramin combines with proteins, especially trypanosomal glycolytic enzymes, to inhibit energy metabolism.[Moore TA (2015). “246e: Agents Used to Treat Parasitic Infections”. In Kasper DL, et al. (eds.). Harrison’s Principles of Internal Medicine (19th ed.). McGraw-Hil. ISBN 9780071802161.]
- History – Suramin was first made by the chemists Oskar Dressel, Richard Kothe and Bernhard Heymann at Bayer AG laboratories in Elberfeld, after research on a series of urea-like compounds. The drug is still sold by Bayer under the brand name Germanin. The chemical structure of suramin was kept secret by Bayer for commercial and strategic reasons, but it was elucidated and published in 1924 by Ernest Fourneau and his team at the Pasteur Institute.[Sneader W (2005). Drug Discovery: A History. John Wiley & Sons. ISBN 9780471899792.]: 378–379 [Fourneau E, Théfouël VJ, Vallée J (1924). “Sur une nouvelle série de médicaments trypanocides”. Comptes Rendus des Séances de l’Académie des Sciences. 178: 675.]
- Research – It is also used as a research reagent to inhibit the activation of heterotrimeric G proteins in a variety of GPCRs with varying potency. It prevents the association of heteromeric G proteins and therefore the receptors guanine exchange functionality (GEF). With this blockade the GDP will not release from the Gα subunit so it cannot be replaced by a GTP and become activated. This has the effect of blocking downstream G protein mediated signaling of various GPCR proteins including rhodopsin, the A1 adenosine receptor, the D2 receptor,[Beindl W, Mitterauer T, Hohenegger M, Ijzerman AP, Nanoff C, Freissmuth M (August 1996). “Inhibition of receptor/G protein coupling by suramin analogues”. Molecular Pharmacology. 50 (2): 415–423. PMID 8700151. Archived from the original on 8 September 2017.] the P2 receptor,[Abbracchio MP, Burnstock G, Boeynaems JM, Barnard EA, Boyer JL, Kennedy C, et al. (September 2006). “International Union of Pharmacology LVIII: update on the P2Y G protein-coupled nucleotide receptors: from molecular mechanisms and pathophysiology to therapy”. Pharmacological Reviews. 58 (3): 281–341. doi:10.1124/pr.58.3.3. PMC 3471216. PMID 16968944.][Khakh BS, Burnstock G, Kennedy C, King BF, North RA, Séguéla P, et al. (March 2001). “International union of pharmacology. XXIV. Current status of the nomenclature and properties of P2X receptors and their subunits”. Pharmacological Reviews. 53 (1): 107–118. PMID 11171941. Archived from the original on 18 November 2016.] and ryanodine receptors.[Wolner I, Kassack MU, Ullmann H, Karel A, Hohenegger M (October 2005). “Use-dependent inhibition of the skeletal muscle ryanodine receptor by the suramin analogue NF676”. British Journal of Pharmacology. 146 (4): 525–533. doi:10.1038/sj.bjp.0706359. PMC 1751178. PMID 16056233.]. Suramin is also an inhibitor of ABC-type [Buxbaum, E.: Co-operative binding sites for transported substrates in the multiple drug resistance transporter Mdr1. Eur. J. Biochem. 265:1 (2001) 64-70 doi:10.1046/j.1432-1327.1999.00644.x] and P-type [Fortes, P.A.G., Ellory, J.C., & Lew, V.L.: Suramin: a potent ATPase inhibitor which acts on the inside surface of the sodium pump. Biochim. Biophys. Acta 318:2 (1973) 262-272, doi:10.1016/0005-2736(73)90119-3.] ATPases, which acts competitively with ATP.
- Suramin was studied as a possible treatment for prostate cancer in a clinical trial.[Ahles TA, Herndon JE, Small EJ, Vogelzang NJ, Kornblith AB, Ratain MJ, et al. (November 2004). “Quality of life impact of three different doses of suramin in patients with metastatic hormone-refractory prostate carcinoma: results of Intergroup O159/Cancer and Leukemia Group B 9480”. Cancer. 101 (10): 2202–2208. doi:10.1002/cncr.20655. PMID 15484217. S2CID 29107328.]
- Suramin has been studied in a mouse model of autism and in a small phase I/II human trial.[LaFee S, Buschman H (26 May 2017). “Researchers Studying Century-Old Drug in Potential New Approach to Autism”. UC San Diego Health. Archived from the original on 1 June 2017.][Naviaux JC, Schuchbauer MA, Li K, Wang L, Risbrough VB, Powell SB, et al. (June 2014). “Reversal of autism-like behaviors and metabolism in adult mice with single-dose antipurinergic therapy”. Translational Psychiatry. 4 (6): e400. doi:10.1038/tp.2014.33. PMC 4080315. PMID 24937094.][Naviaux RK, Curtis B, Li K, Naviaux JC, Bright AT, Reiner GE, et al. (July 2017). “Low-dose suramin in autism spectrum disorder: a small, phase I/II, randomized clinical trial”. Annals of Clinical and Translational Neurology. 4 (7): 491–505. doi:10.1002/acn3.424. PMC 5497533. PMID 28695149.][“Q and A – Suramin and Autism”. UC Health – UC San Diego. Retrieved 27 July 2021.] Results from a randomized clinical study found no statistically significant effects of suramin (in either 10mg or 20mg doses) versus placebo on boys with moderate to severe autism spectrum disorder.[Hough D, Mao AR, Aman M, Lozano R, Smith-Hicks C, Martinez-Cerdeno V, et al. (November 2023). “Randomized clinical trial of low dose suramin intravenous infusions for treatment of autism spectrum disorder”. Annals of General Psychiatry. 22 (1): 45. doi:10.1186/s12991-023-00477-8. PMC 10626700. PMID 37932739.]
- Suramin is a reversible and competitive protein-tyrosine phosphatase (PTPases) inhibitor, also is the potent inhibitor of sirtuins, purified topoisomerase II and SARS-CoV-2 RNA-dependent RNA polymerase (RdRp).[“Suramin sodium salt”. Selleck Chemicals.]
- Further reading: Zhang YL, Keng YF, Zhao Y, Wu L, Zhang ZY (May 1998). “Suramin is an active site-directed, reversible, and tight-binding inhibitor of protein-tyrosine phosphatases”. The Journal of Biological Chemistry. 273 (20): 12281–12287. doi:10.1074/jbc.273.20.12281. PMID 9575179. all these notes from Suramin – Wikipedia
- Xanthines like caffeine and pentifylline activate it by potentiating sensitivity to native ligand Ca.
- Physiological agonist: Cyclic ADP-ribose can act as a physiological gating agent. It has been suggested that it may act by making FKBP12.6 (12.6 kilodalton FK506 binding protein, as opposed to 12 kDa FKBP12 which binds to RyR1) which normally bind (and blocks) RyR2 channel tetramer in an average stoichiometry of 3.6, to fall off RyR2 (which is the predominant RyR in pancreatic beta cells, cardiomyocytes and smooth muscles).[13]
- Agonist: 4-chloro-m-cresol and suramin are direct agonists, i.e., direct activators.
A variety of other molecules may interact with and regulate ryanodine receptor. For example: dimerized Homer physical tether linking inositol trisphosphate receptors (IP3R) and ryanodine receptors on the intracellular calcium stores with cell surface group 1 metabotropic glutamate receptors and the Alpha-1D adrenergic receptor[14]
Ryanodine
The plant alkaloid ryanodine, for which this receptor was named, has become an invaluable investigative tool. It can block the phasic release of calcium, but at low doses may not block the tonic cumulative calcium release. The binding of ryanodine to RyRs is use-dependent, that is the channels have to be in the activated state. At low (<10 micromolar, works even at nanomolar) concentrations, ryanodine binding locks the RyRs into a long-lived subconductance (half-open) state and eventually depletes the store, while higher (~100 micromolar) concentrations irreversibly inhibit channel-opening.
Caffeine
RyRs are activated by millimolar caffeine concentrations. High (greater than 5 mmol/L) caffeine concentrations cause a pronounced increase (from micromolar to picomolar) in the sensitivity of RyRs to Ca2+ in the presence of caffeine, such that basal Ca2+ concentrations become activatory. At low millimolar caffeine concentrations, the receptor opens in a quantal way, but has complicated behavior in terms of repeated use of caffeine or dependence on cytosolic or luminal calcium concentrations.
Role in disease
RyR1 mutations are associated with malignant hyperthermia and central core disease.[15] Mutant-type RyR1 receptors exposed to volatile anesthetics or other triggering agents can display an increased affinity for cytoplasmic Ca2+ at activating sites as well as a decreased cytoplasmic Ca2+ affinity at inhibitory sites.[16] The breakdown of this feedback mechanism causes uncontrolled release of Ca2+ into the cytoplasm, and increased ATP hydrolysis resulting from ATPase enzymes shuttling Ca2+ back into the sarcoplasmic reticulum leads to excessive heat generation.[17]
RyR2 mutations play a role in stress-induced polymorphic ventricular tachycardia (a form of cardiac arrhythmia) and ARVD.[2] It has also been shown that levels of type RyR3 are greatly increased in PC12 cells overexpressing mutant human Presenilin 1, and in brain tissue in knockin mice that express mutant Presenilin 1 at normal levels,[18] and thus may play a role in the pathogenesis of neurodegenerative diseases, like Alzheimer’s disease.[19]
The presence of antibodies against ryanodine receptors in blood serum has also been associated with myasthenia gravis (i.e., MG).[1] Individuals with MG who have antibodies directed against ryanodine receptors typically have a more severe form of generalized MG in which their skeletal muscle weaknesses involve muscles that govern basic life functions.[20]
Sudden cardiac death in several young individuals in the Amish community (four of which were from the same family) was traced to homozygous duplication of a mutant RyR2 (Ryanodine Receptor) gene.[21] Normal (wild type) ryanodine receptors are involved in CICR in heart and other muscles, and RyR2 functions primarily in the myocardium (heart muscle).
Structure
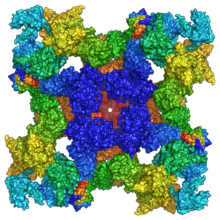
Ryanodine receptors are multidomain homotetramers which regulate intracellular calcium ion release from the sarcoplasmic and endoplasmic reticula.[22] They are the largest known ion channels, with weights exceeding 2 megadaltons, and their structural complexity enables a wide variety of allosteric regulation mechanisms.[23][24]
RyR1 cryo-EM structure revealed a large cytosolic assembly built on an extended α-solenoid scaffold connecting key regulatory domains to the pore. The RyR1 pore architecture shares the general structure of the six-transmembrane ion channel superfamily. A unique domain inserted between the second and third transmembrane helices interacts intimately with paired EF-hands originating from the α-solenoid scaffold, suggesting a mechanism for channel gating by Ca2+.[1][25]
See also
- Ryanoid, a class of insecticide that act through ryanodine receptors
- Neuromuscular junction
- Dihydropyridine receptor
References
- Santulli G, Marks AR (2015). “Essential Roles of Intracellular Calcium Release Channels in Muscle, Brain, Metabolism, and Aging”. Current Molecular Pharmacology. 8 (2): 206–222. doi:10.2174/1874467208666150507105105. PMID 25966694.
- Zucchi R, Ronca-Testoni S (March 1997). “The sarcoplasmic reticulum Ca2+ channel/ryanodine receptor: modulation by endogenous effectors, drugs and disease states”. Pharmacological Reviews. 49 (1): 1–51. PMID 9085308.
- Fabiato A (July 1983). “Calcium-induced release of calcium from the cardiac sarcoplasmic reticulum”. The American Journal of Physiology. 245 (1): C1-14. doi:10.1152/ajpcell.1983.245.1.C1. PMID 6346892.
- Cheng H, Lederer WJ, Cannell MB (October 1993). “Calcium sparks: elementary events underlying excitation-contraction coupling in heart muscle”. Science. 262 (5134): 740–744. Bibcode:1993Sci…262..740C. doi:10.1126/science.8235594. PMID 8235594.
- Bround MJ, Wambolt R, Luciani DS, Kulpa JE, Rodrigues B, Brownsey RW, et al. (June 2013). “Cardiomyocyte ATP production, metabolic flexibility, and survival require calcium flux through cardiac ryanodine receptors in vivo”. The Journal of Biological Chemistry. 288 (26): 18975–18986. doi:10.1074/jbc.M112.427062. PMC 3696672. PMID 23678000.
- Tsuboi T, da Silva Xavier G, Holz GG, Jouaville LS, Thomas AP, Rutter GA (January 2003). “Glucagon-like peptide-1 mobilizes intracellular Ca2+ and stimulates mitochondrial ATP synthesis in pancreatic MIN6 beta-cells”. The Biochemical Journal. 369 (Pt 2): 287–299. doi:10.1042/BJ20021288. PMC 1223096. PMID 12410638.
- Dror V, Kalynyak TB, Bychkivska Y, Frey MH, Tee M, Jeffrey KD, et al. (April 2008). “Glucose and endoplasmic reticulum calcium channels regulate HIF-1beta via presenilin in pancreatic beta-cells”. The Journal of Biological Chemistry. 283 (15): 9909–9916. doi:10.1074/jbc.M710601200. PMID 18174159.
- Meissner G, Darling E, Eveleth J (January 1986). “Kinetics of rapid Ca2+ release by sarcoplasmic reticulum. Effects of Ca2+, Mg2+, and adenine nucleotides”. Biochemistry. 25 (1): 236–244. doi:10.1021/bi00349a033. PMID 3754147.
- Van Petegem F (September 2012). “Ryanodine receptors: structure and function”. The Journal of Biological Chemistry. 287 (38): 31624–31632. doi:10.1074/jbc.r112.349068. PMC 3442496. PMID 22822064.
- Handhle A, Ormonde CE, Thomas NL, Bralesford C, Williams AJ, Lai FA, Zissimopoulos S (November 2016). “Calsequestrin interacts directly with the cardiac ryanodine receptor luminal domain”. Journal of Cell Science. 129 (21): 3983–3988. doi:10.1242/jcs.191643. PMC 5117208. PMID 27609834.
- Vites AM, Pappano AJ (March 1994). “Distinct modes of inhibition by ruthenium red and ryanodine of calcium-induced calcium release in avian atrium”. The Journal of Pharmacology and Experimental Therapeutics. 268 (3): 1476–1484. PMID 7511166.
- Xu L, Tripathy A, Pasek DA, Meissner G (September 1998). “Potential for pharmacology of ryanodine receptor/calcium release channels”. Annals of the New York Academy of Sciences. 853 (1): 130–148. Bibcode:1998NYASA.853..130T. doi:10.1111/j.1749-6632.1998.tb08262.x. PMID 10603942. S2CID 86436194.
- Wang YX, Zheng YM, Mei QB, Wang QS, Collier ML, Fleischer S, et al. (March 2004). “FKBP12.6 and cADPR regulation of Ca2+ release in smooth muscle cells”. American Journal of Physiology. Cell Physiology. 286 (3): C538–C546. doi:10.1152/ajpcell.00106.2003. PMID 14592808. S2CID 20900277.
- Tu JC, Xiao B, Yuan JP, Lanahan AA, Leoffert K, Li M, et al. (October 1998). “Homer binds a novel proline-rich motif and links group 1 metabotropic glutamate receptors with IP3 receptors”. Neuron. 21 (4): 717–726. doi:10.1016/S0896-6273(00)80589-9. PMID 9808459. S2CID 2851554.
- Robinson RL, Brooks C, Brown SL, Ellis FR, Halsall PJ, Quinnell RJ, et al. (August 2002). “RYR1 mutations causing central core disease are associated with more severe malignant hyperthermia in vitro contracture test phenotypes”. Human Mutation. 20 (2): 88–97. doi:10.1002/humu.10098. PMID 12124989. S2CID 21497303.
- Yang T, Ta TA, Pessah IN, Allen PD (July 2003). “Functional defects in six ryanodine receptor isoform-1 (RyR1) mutations associated with malignant hyperthermia and their impact on skeletal excitation-contraction coupling”. The Journal of Biological Chemistry. 278 (28): 25722–25730. doi:10.1074/jbc.m302165200. PMID 12732639.
- Reis M, Farage M, de Meis L (2002-01-01). “Thermogenesis and energy expenditure: control of heat production by the Ca(2+)-ATPase of fast and slow muscle”. Molecular Membrane Biology. 19 (4): 301–310. doi:10.1080/09687680210166217. PMID 12512777. S2CID 10720335.
- Chan SL, Mayne M, Holden CP, Geiger JD, Mattson MP (June 2000). “Presenilin-1 mutations increase levels of ryanodine receptors and calcium release in PC12 cells and cortical neurons”. The Journal of Biological Chemistry. 275 (24): 18195–18200. doi:10.1074/jbc.M000040200. PMID 10764737.
- Gong S, Su BB, Tovar H, Mao C, Gonzalez V, Liu Y, et al. (June 2018). “Polymorphisms Within RYR3 Gene Are Associated With Risk and Age at Onset of Hypertension, Diabetes, and Alzheimer’s Disease”. American Journal of Hypertension. 31 (7): 818–826. doi:10.1093/ajh/hpy046. PMID 29590321.
- Gilhus NE (July 2023). “Myasthenia gravis, respiratory function, and respiratory tract disease”. Journal of Neurology. 270 (7): 3329–3340. doi:10.1007/s00415-023-11733-y. PMC 10132430. PMID 37101094.
- Tester DJ, Bombei HM, Fitzgerald KK, Giudicessi JR, Pitel BA, Thorland EC, et al. (March 2020). “Identification of a Novel Homozygous Multi-Exon Duplication in RYR2 Among Children With Exertion-Related Unexplained Sudden Deaths in the Amish Community”. JAMA Cardiology. 5 (3): 13–18. doi:10.1001/jamacardio.2019.5400. PMC 6990654. PMID 31913406.
- Santulli G, Lewis D, des Georges A, Marks AR, Frank J (2018). “Ryanodine Receptor Structure and Function in Health and Disease”. In Harris JR, Boekema EJ (eds.). Membrane Protein Complexes: Structure and Function. Vol. 87. Singapore: Springer Singapore. pp. 329–352. doi:10.1007/978-981-10-7757-9_11. ISBN 978-981-10-7756-2. PMC 5936639. PMID 29464565.
{{cite book}}:|journal=ignored (help) - Lanner JT, Georgiou DK, Joshi AD, Hamilton SL (November 2010). “Ryanodine receptors: structure, expression, molecular details, and function in calcium release”. Cold Spring Harbor Perspectives in Biology. 2 (11): a003996. doi:10.1101/cshperspect.a003996. PMC 2964179. PMID 20961976.
- Van Petegem F (January 2015). “Ryanodine receptors: allosteric ion channel giants”. Journal of Molecular Biology. 427 (1): 31–53. doi:10.1016/j.jmb.2014.08.004. PMID 25134758.
- Zalk R, Clarke OB, des Georges A, Grassucci RA, Reiken S, Mancia F, et al. (January 2015). “Structure of a mammalian ryanodine receptor”. Nature. 517 (7532): 44–49. Bibcode:2015Natur.517…44Z. doi:10.1038/nature13950. PMC 4300236. PMID 25470061.
External links
- Ryanodine+Receptor at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
Leave a Reply